 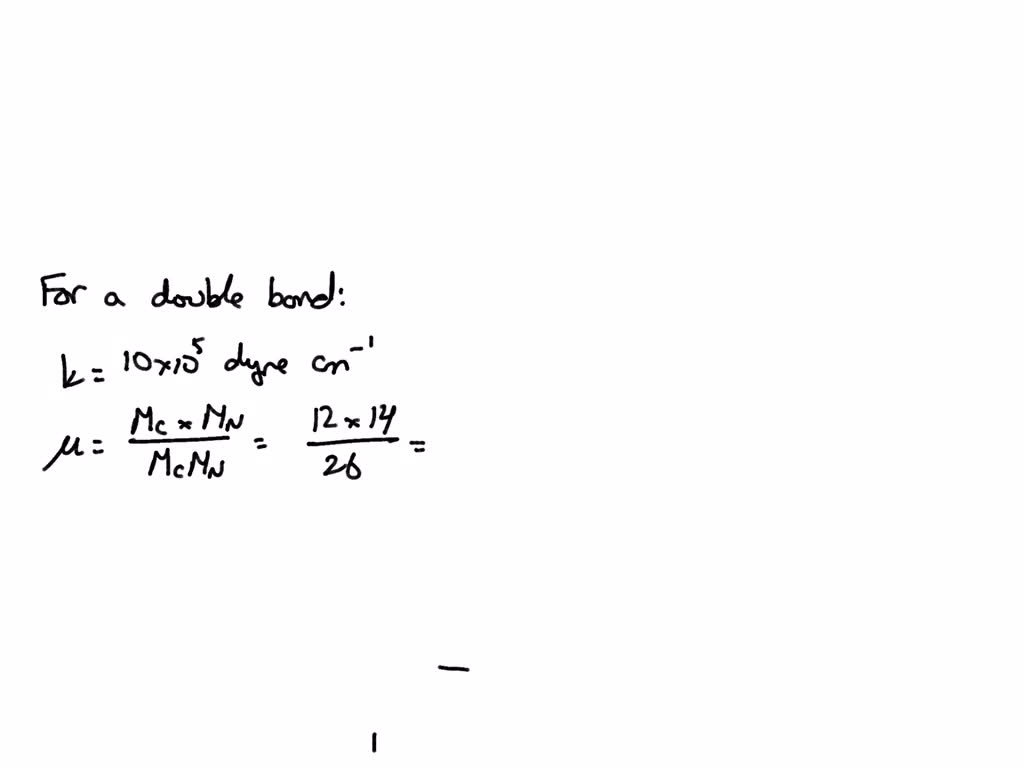
Note: Although Na 2CO 3 is an ionic compound and sodium is present in an ionic form, it is a habitual way of referring to an element in these types of calculations. This is the accurate way of rounding off to the correct number of significant figures in multistep calculations. I do it here mainly to save space, but you can keep the numbers in the calculator and round them off at the end. To use the calculator, simply input the chemical formula of the substance and click the 'Calculate' button. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. Note that no atom in our hypothetical element has a mass of 10. The Molar Mass Calculator is a tool used to calculate the molar mass of a substance based on its chemical formula. The molar mass of a substance ( M) is the weight of one mole of the substance. Fe4 Fe (CN)63, NaHCO3, ch3coonh4, h2so4, pb (c2h3o2)23h2o, caso41/2h2o) and press Enter or click Calculate button. In this example, we have the following: 0.50 × 10 u. Enter a chemical formula to calculate its molar mass (e.g. The little inconsistency in the final answer is due to rounding off the intermediate numbers. The sum is the weighted average and serves as the formal atomic mass of the element. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12.011 g/mol: Table 2.4.1 2.4. So, to calculate the moles from a given mass, we make a conversion factor correlating 1 mole with the molar of the given component.įor example, how many moles of sulfur are there in a 16.2 g sample? The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. The molar mass, on the other hand, is a constant number for a given atom or a molecule as it is for a specific amount of it. The given mass is the mass of the sample, and it can be any number, for example, we can have 10 g of salt, 15 g, or 100 g. To calculate the moles from a given mass (m), the molar mass of the component is used. The molecular mass of water is 18.0 amu, and the molar mass is 18.0 g/mol.įor the molar mass, keeping one decimal is usually an acceptable approximation, and for the Avogadro’s number, you can use 6.02 x 10 23. 207.2 Pb Lead 114 (289) Uuq Ununquadium 10 20.1797 Ne Neon 18 39.948 Ar Argon 36 83.798 Kr Krypton 54 131.293 Xe Xenon 86 (222) Rn Radon 118 (294) Uuo Ununoctium 2 4.002 60 He Helium 57 138.905 47 La Lanthanum 58 140.116 Ce Cerium 59 140.907 65 Pr Praseodymium 60 144.242 Nd Neodymium 61 (145) Pm Promethium 62 150.36 Sm Samarium 63 151.964 Eu. This 118 element printable periodic table is perfect for kids or anyone who wants a clean-looking chart with atomic weights rounded to the usual two decimal points. Numerically, the molar mass is equal to the atomic mass of a given atom or a molecule, so we can look up the molar mass of an element in the periodic table.įor example, we can say that the mass of Cu is 63.55 amu or 63.55 g/mol. The mass of one mole of atoms/molecules/ions is called its molar mass (M) expressed in g/mol. One mole of any element contains 6.022 x 10 23 of its atoms, and a mole of a compound contains 6.022 x 10 23 molecules, ions-ions, and in general, 1 mol = 6.022 x 10 23 particles. This is called Avogadro’s number ( N A = 6.022 x 10 23). By definition, a mole is the number of atoms in a 12-g sample of 12C isotope which happens to be 6.022 x 10 23 atoms. High-pressure SF 6 gas is used in place of older oils that may have contaminants that are not environmentally friendly (part (c) in the accompanying figure).The mole is just a number like a dozen, a hundred, or a million. Sulfur hexafluoride also has another interesting use: a spark suppressant in high-voltage electrical equipment. (c) A high-voltage electrical switchgear assembly that would be filled with SF 6 as a spark suppressant. 
Thus, the molar mass of bilirubin can be expressed as 584.05 g/mol, which is read as five hundred eighty four. The division sign (/) implies per, and 1 is implied in the denominator. Using a Periodic Table with Two Decimal Places, calculate the Molar Mass of Sr (CIO), with correct Significant Figures. (a) Properly protected workers clear out the Tokyo subway after the nerve toxin sarin was released. Because molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). 207.2 Pb Lead 114 (289) Uuq Ununquadium 10 20.1797 Ne Neon 18 39.948 Ar Argon 36 83.798 Kr Krypton 54 131. \): Sarin and Sulfur Hexafluoride © Thinkstock. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
